We use cookies to improve your experience. By accepting you agree to our cookie policy

Neuromodulation is a form of treatment that uses small electrical devices placed inside the body to help manage pain. These devices can gently stimulate nerves, the spinal cord, or specific areas that carry pain signals, which may help reduce the pain you feel.
There are many versions of neuromodulators to help manage Complex Regional Pain Syndrome (CRPS) such as Spinal Cord Stimulators (SCS), Dorsal Root Ganglion Stimulators, Peripheral Nerve Stimulators plus Transcranial Stimulators.
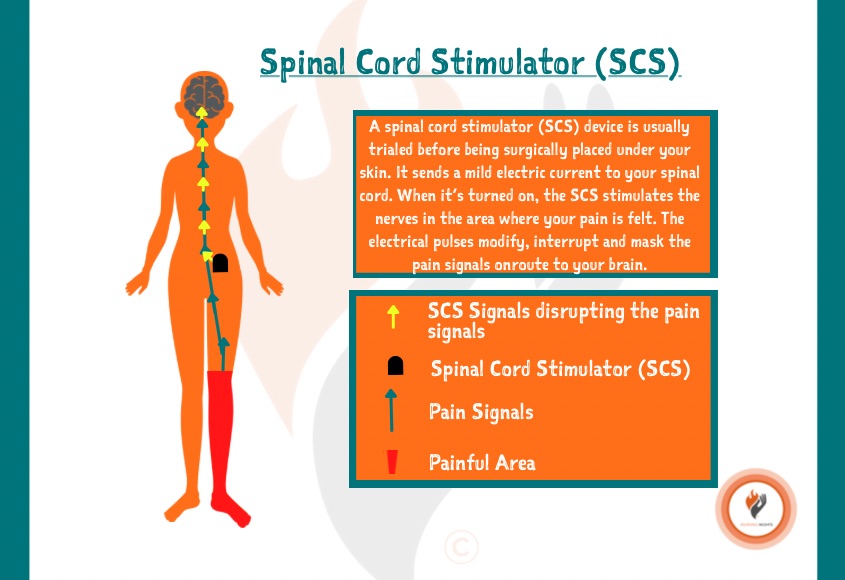
Spinal Cord Stimulation (SCS) treatment is seen as an invasive treatment option for CRPS. It is also an expensive treatment and many PCT's / hospitals are reluctant to do this. The cost is in the region of £15,000 on the NHS and so you would usually have a trial stimulator put in for a several days, a couple of weeks or a month and then if the trial is successful for you in reducing your pain and symptoms, they will implant the full SCS.
Before you are considered for an SCS, you have to go through a full psychological assessment. This psychological assessment is an important part of the process of implanting an SCS. This is because it has been suggested that psychological and social factors can influence the outcome of the procedure.
Electrodes are put on the spinal cord and a battery pack is inserted at the front of your body under your skin in the abdomen region. You are given a 'wand' to place on the outside where the battery pack is and you can control the on/off and volume of the stimulation up and down.
There are now different types of SCS, including a 10 kHz High Frequency SCS (Russo, M & Van Buyten, J-P. 2015). Usually, the SCS is seen and implanted as a last resort treatment due to various implications, including the cost and invasiveness of the procedure However, research studies, such as the Poree, L. et al. (2012) study, say that: "Newer information regarding safety, cost, and efficacy leads us to believe that SCS for the treatment of CRPS should be implemented earlier in a treatment algorithm using a more comprehensive approach."
Spinal Cord Stimulation is considered favourable and effective management for CRPS and has high-level evidence showing that SCS can improve patients' pain, quality of life and pain score. (Visnjevac O et al 2017)
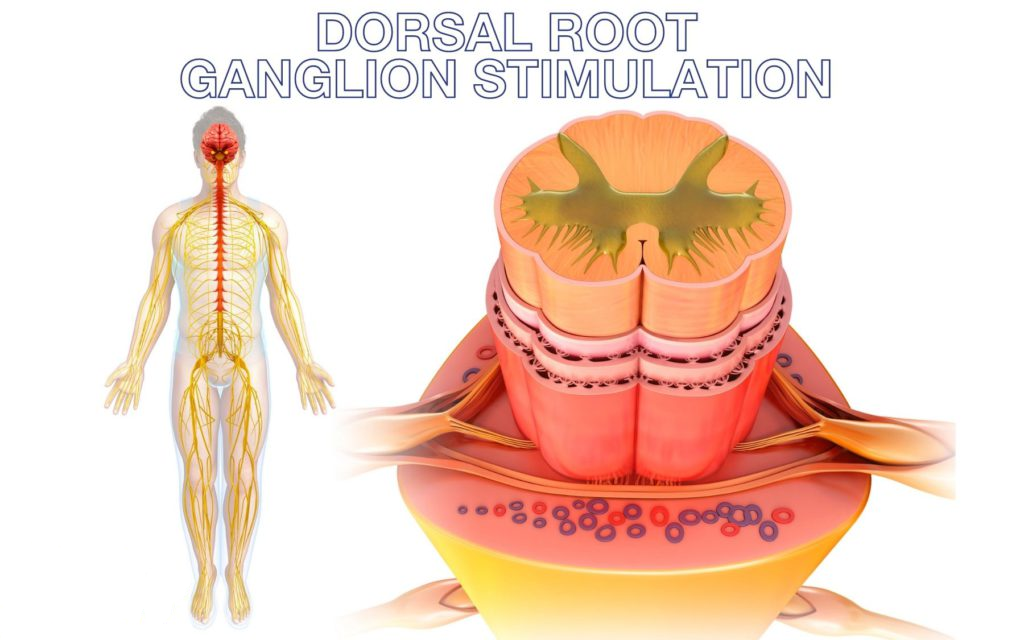
The Dorsal Root Ganglion (DRG) is a small bundle of nerves that are involved in sending pain messages to the brain. They control when sensations can enter your spinal cord and you will find there is a DRG in every section of bone in your spine. DRG stimulation uses implanted equipment under your skin in your spine to send mild electrical impulses to the area of the DRG in your spine.
DRG stimulation is a newer form of treatment than the SCS, however you would still need to go through the psychological assessment. In February 2024, the network of Psychologists in Pain Neuromodulation developed a document called 'Good practice guidelines for psychological assessment and intervention for pain neuromodulation services' to give the sector some guidance in assessing people going through the operation.
For more information about this treatment, read our Complete Guide To DRG Stimulation Treatment article. You can download our 24-page guide to Dorsal Root Ganglion stimulation for CRPS booklet or purchase a physical copy from the Burning Nights CRPS Support Shop.

We use cookies to improve your experience. By accepting you agree to our cookie policy
 £
£