We use cookies to improve your experience. By accepting you agree to our cookie policy

Thank you Dr. Morland for agreeing to explain research for CRPS and wanting to help raise awareness for this condition by authoring this series of blogs “Research Studies Explained!”
In the last 20 years, research into Complex Regional Pain Syndrome (CRPS) has seen huge advances, taking it from a little understood and assumed-rare condition, to the realisation that it is an incredibly complex disorder that may in fact describe a whole group of related pain conditions.
Complex Regional Pain Syndrome or CRPS often develops after a seemingly minor injury, which instead of healing normally triggers an over-reaction of pain and inflammation systems in the body.
The Budapest criteria are often used to diagnose CRPS. These look at four main categories of symptoms as shown below:
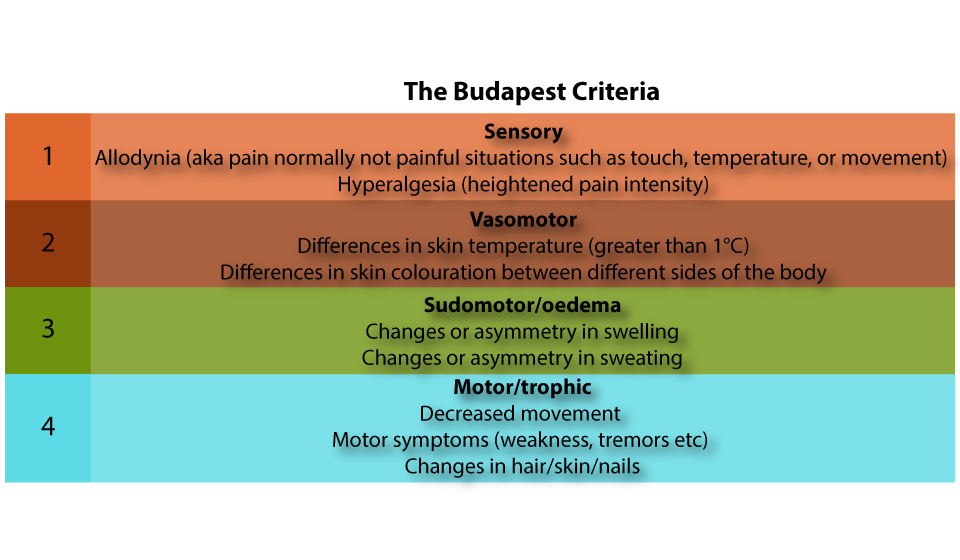
These symptoms are used to identify CRPS according to the following checklist:
A: Ongoing pain at an intensity which cannot be explained by the triggering event (e.g. a fall or fracture)
B: at least one sign (i.e. measured experience) from two (2) or more of the four (4) categories above
C: at least one symptom (i.e. reported experience) from three or more of the four categories above
D: A lack of alternative diagnosis
 This last point highlights the difficulties both doctors and researchers face when trying to develop animals models of CRPS.
This last point highlights the difficulties both doctors and researchers face when trying to develop animals models of CRPS.
Animal models can be incredibly informative when trying to understanding how painful conditions develop – they have been used to identify changes that occur at the cellular level in pain, helping to understand the changes that take place when pain changes from acute (useful & teaches us to avoid the dangerous things in life), to chronic (pain that just won’t go away).
So far, pain researchers have found that the way the body reacts to pain depends on what caused the pain, and also to some extent on individual factors such as genetics, previous life experience, and lifestyle.
When developing animal models, the researcher must first establish that what they are doing is a valid and accurate representation of the human condition.
This applies to both how the model is induced (i.e. what causes the condition), and what signs/symptoms can be detected. For CRPS, this situation is complicated by a lack of understanding of what causes the disease, huge variation in how patients experience the condition, and a reliance on reported CRPS symptoms. For researchers trying to develop accurate models, reported symptoms are the greatest challenge.
In CRPS, animal models usually take one of two forms:
Most models are in this first category, and are based on evidence that CRPS develops following a relatively minor accident, such as a fall resulting in broken skin and/or bones.
Such injuries affect the body in a number of different ways, and so are best looked at by breaking them down into elements, such as the effect of a fracture and subsequent bed-rest (‘immobilisation’); how nerves change the way they transmit pain signals when they are crushed by swelling, fractures, or other injuries; and how damage can happen when the blood supply is restored to an injured limb.
Together, these models can identify how each different element of an injury contribute to the symptoms experienced.
As the immune system is incredibly complex, and it has been difficult to identify a unique “signature” for the immune response in CRPS, there are fewer researchers looking at the immune aspects of CRPS.
However, a recent study found that disrupting the activity of a certain type of immune cell (B cells) in mice decreases CRPS-like behaviours such as pain and negative vascular changes following a fracture.
This suggests that being able to control the immune reaction could decrease the chances of developing CRPS following an injury. Other studies have looked at the effect of nerve inflammation, as present following a minor injury, and what factors are responsible for the transition from normal immune response to injury, and the uncontrolled response immune seen in CRPS.
 Another recent study looked at the role of the immune system from a different angle, by injecting serum from CRPS patients into mice. Samples from CRPS patients have been shown to have high levels of inflammation, and when this was injected into mice, they showed CRPS-like symptoms that not seen in mice injected with serum from healthy volunteers.
Another recent study looked at the role of the immune system from a different angle, by injecting serum from CRPS patients into mice. Samples from CRPS patients have been shown to have high levels of inflammation, and when this was injected into mice, they showed CRPS-like symptoms that not seen in mice injected with serum from healthy volunteers.
This suggests there is something different in the serum of CRPS patients that could explain the different reaction to injury. However, in this study, the patient group was selected to be similar, and as CRPS can present in a wide variety of ways, these results are only relevant to that specific patient group. Studies are already in progress to try and link what is seen in serum to specific symptoms experienced, so in the years to come, we can expect a lot more work like this.
Once a model has been made, the researcher must then find ways of testing for the signs/symptoms reported by patients. Not all of these are easy to detect in animals, pain being one of the most difficult. Most methods of measuring pain in animals look only at hypersensitivity.
Hypersensitivity, or the perception of pain greater than would be expected, can be measured in animals by looking at how different models change responses to increasing temperature (up to 48°C), and increasing force (using a hair-like instrument – von Frey Hairs).
However, pain is not just a sensory experience, and is always associated with emotional symptoms, which are just as damaging to the sufferer, and much more difficult to measure in animals. To study this aspect of pain, researchers look at changes in the natural behaviour of the animal, such as how readily they explore a new space (theory: pain decreases the perceived risk of exploration), and also how they react to other animals (theory: animals in pain behave differently around other animals based on whether they are familiar or a risk).
It is very important when studying pain to ensure that any treatments developed tackle both the sensory and emotional aspects of pain.
As discussed, most models used to study CRPS are limited in their application, as they focus on a very specific set of conditions, such as bone fracture models only being applicable to CRPS patients who developed the condition via fractures, or the immune serum study only applying to patients which fit the same profile.
By looking at a range of different symptoms of CRPS, and how they compare across different models, researchers should be able to build up a detailed picture of what factors contribute to each symptom and how they can be combatted.
It is important to recognise that advances in CRPS research are reliant on identifying the biological changes responsible for the symptoms of CRPS, and without a definitive cause the only way to do this is to look at a range of different models.
As we learn more about the processes happening in the body that are responsible for each symptom, and how they change during disease progression, we get closer to developing useful treatments that take into account all the different ways CRPS patients experience the condition.
The personal nature of the pain experience, combined with the variation in symptoms experienced in CRPS mean there is never, alas, going to be a one-size-fits-all treatment, but with greater understanding, diagnoses could become more accurate, and appropriate treatments, based on the unique symptom profile of the patient could become a reality.
 Some interesting open access articles on models of CRPS:
Some interesting open access articles on models of CRPS:Linnman, C et al. (2013) ‘Inflaming the brain: CRPS a model disease to understand neuroimmune interactions in chronic pain,’ Journal of Neuroimmune Pharmacology & NCBI NIH. June 2013. Vol 8 (3) pp 547-563. Available from: < http://www.ncbi.nlm.nih.gov/pubmed/23188523> doi: 10.1007/s11481-012-9422-8
Cooper, M.S., Clark, V.P. (2013) ‘Neuroinflammation, neuroautoimmunity, and the co-morbidities of complex regional pain syndrome,’ Journal of Neuroimmune Pharmacology & NCBI NIH. June 2013. Vol 8 (3), pp 452-469. Available from: <http://www.ncbi.nlm.nih.gov/pubmed/22923151> doi: 10.1007/s11481-012-9392-x
National Centre for the Replacement & Reduction of Animals in Research – Available from: <https://www.nc3rs.org.uk/>
Rosie recently completed a PhD in neuroscience and pain studies from Imperial College, and has a special interest in how animal models can help increase understanding of complex pain disorders. Visit ROSIE’S BLOG for more details for more details.
THANK YOU Dr. MORLAND for an excellent piece of Research Studies Explained and how animal models are developed of CRPS.
Please contact Burning Nights if you have a topic you wish Dr. Morland to explain or a piece of research you wish explained! Feel free to share this post or any other posts on Social Media or make a comment here below!
Last Updated: 18/01/2020
We use cookies to improve your experience. By accepting you agree to our cookie policy
 £
£